Mumbai (PTI): The Bombay High Court on Wednesday ordered for fresh testing of Johnson & Johnson baby powder samples and permitted the company to manufacture the product but to not sell it, as per the Maharashtra government order.
The company had filed a petition challenging two orders of the state government- one dated September 15 cancelling the license and the second dated September 20 ordering to immediately stop manufacturing and sale of the company's baby powder product.
The orders were passed by the joint commissioner and licensing authority of the state Food and Drug Administration (FDA).
The government based its orders on the report of the Central Drug Laboratory in Kolkata, which found the powder containing higher pH level than prescribed.
A division bench of Justices S V Gangapurwala and S G Dige on Wednesday directed the FDA to collect fresh samples from the company's factory in Mulund area of Mumbai within three days.
The sample would then be sent to three laboratories - two government labs and a private one - for testing.
"The samples shall be sent to the Central Drug Testing Laboratory, west zone, FDA lab and the Intertech Laboratory for testing," the court said.
"The labs shall submit a report within one week thereafter," it added.
Senior counsel Ravi Kadam, appearing for the company, sought that the court till then permit the company to at least start manufacturing the product.
"The petitioner is restrained from selling or distributing the baby powder product by the government. The company shall abide by this order. If the company chooses to manufacture the product then it would be at their own risk," the bench said.
The HC posted the matter for further hearing on November 30.
The company had in its plea said 14 random batches of February, March and September 2022 were tested by an independent public testing laboratory and all were found to be well within the prescribed pH value.
The leading Fast Moving Consumer Goods (FMCG) manufacturer said it has been making the baby powder at its Mulund plant for the last 57 years and its license was renewed in January 2020.
The company also said due to the license revocation, it suffered a daily loss of Rs 2.5 crore based on the market value of the goods sold.
Let the Truth be known. If you read VB and like VB, please be a VB Supporter and Help us deliver the Truth to one and all.
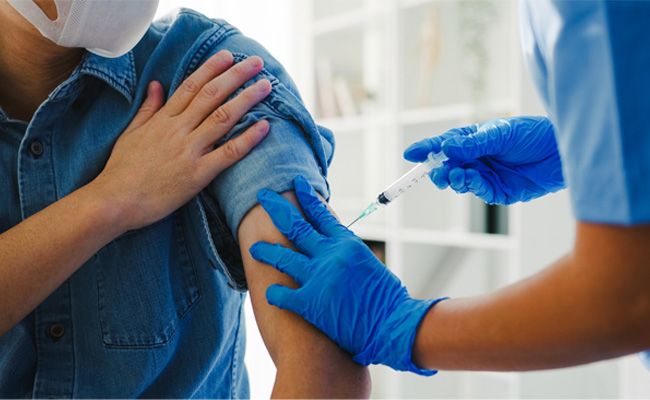
Mumbai (PTI): The Maharashtra government has set up a State Vaccination Task Force to strengthen the regular immunisation programme and review the progress of related campaigns, a health department official said on Monday.
The State Vaccination Task Force will comprise at least 29 members and will be headed by the administrative head of the health department, he informed.
The government has also constituted separate district-level and municipal vaccination task forces to improve implementation and address challenges at the grassroots level, he said.
Municipal task forces, chaired by respective civic commissioners, have been constituted in view of the vast urban population in Maharashtra and the role of civic bodies in implementing different health programmes.
The district-level task forces will function under the chairmanship of collectors.
"Complete immunisation of children at the appropriate age is an extremely simple, cost-effective and highly effective measure to reduce child mortality and the prevalence of diseases among kids. Immunisation is a powerful tool for reducing illness in children," maintained the official.
To ensure full vaccination of all children, the state government implements various campaigns from time to time as per the central government guidelines, he pointed out.
"Active participation and cooperation of other relevant government departments are essential (in making these campaigns successful)," according to the official.
The state-level body will review the regular immunisation programme, associated campaigns and vaccine-preventable diseases in detail. It will also conduct focused assessments of high-risk districts and municipal corporations, including vacancies at district, municipal and sub-district levels, availability of cold chain equipment, resource gaps and training requirements, he noted.
The state task force will review allocation and utilisation of funds for immunisation and ensure timely action by officers concerned based on reports from district and municipal task forces and state-level monitoring mechanisms, the official said.
It will also ensure active coordination and participation of other government departments in immunisation drives, while district and municipal task forces will carry out similar functions at their respective levels, the official added.