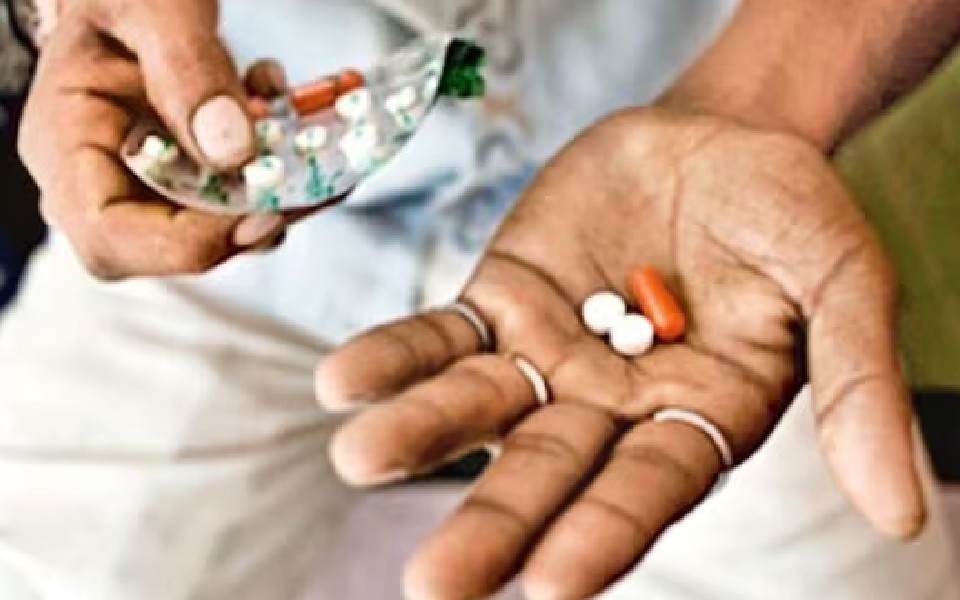
New Delhi, Aug 22: The government has banned 156 widely sold fixed-dose combination (FDC) drugs including antibacterial medicines used for fever, cold, allergies and pain, saying that they are "likely to involve risk to human beings".
FDC drugs are those which contain a combination of two or more active pharmaceutical ingredients in a fixed ratio and are also referred to as "cocktail" drugs.
According to a gazette notification issued by the Union health ministry on August 12, the government has banned 'Aceclofenac 50mg + Paracetamol 125mg tablet', one of the popular combinations used as pain-relieving medicines manufactured by top pharma companies.
The list also includes Mefenamic Acid + Paracetamol Injection, Cetirizine HCl + Paracetamol + Phenylephrine HCl, Levocetirizine + Phenylephrine HCl + Paracetamol, Paracetamol + Chlorpheniramine Maleate + Phenyl Propanolamine and Camylofin Dihydrochloride 25 mg + Paracetamol 300mg.
The Centre also banned the combination of Paracetamol, Tramadol, Taurine and Caffeine. Tramadol is an opioid-based pain killer.
"The Central government is satisfied that the use of the Fixed Dose Combination drug is likely to involve risk to human beings whereas safer alternatives to the said drug are available," the notification said.
It said that the matter was examined by an Expert Committee appointed by the Centre which considered these FDCs as "irrational".
It further stated that the apex panel Drugs Technical Advisory Board (DTAB) also examined these FDCs and recommended that "there is no therapeutic justification for the ingredients contained in these FDCs".
"The FDC may involve risk to human beings. Hence in the larger public interest, it is necessary to prohibit the manufacture, sale or distribution of this FDC under section 26 A of Drugs and Cosmetics Act 1940," the notification read.
"In view of above, any kind of regulation or restriction to allow for any use in patients is not justifiable. Therefore, only prohibition under section 26A is recommended," it added.
Following the DTAB's recommendations, the notification said that "the Central government is satisfied that it is necessary and expedient in public interest to prohibit the manufacture, sale and distribution for human use of the said drug in the country".
The list includes certain products which were already discontinued by many drug makers.
The government in 2016 had announced the ban on the manufacture, sale and distribution of 344 drug combinations after an expert panel, set up at the behest of the Supreme Court, had stated they were being sold to patients without scientific data and the order was challenged by the manufacturers in court, sources said.
In June 2023, 14 FDCs which were part of those 344 drug combinations, were banned. Many of the FDCs banned recently were also from those 344 drug combinations, the sources said.
Let the Truth be known. If you read VB and like VB, please be a VB Supporter and Help us deliver the Truth to one and all.
Mumbai (PTI): The Maharashtra government has set up a State Vaccination Task Force to strengthen the regular immunisation programme and review the progress of related campaigns, a health department official said on Monday.
The State Vaccination Task Force will comprise at least 29 members and will be headed by the administrative head of the health department, he informed.
The government has also constituted separate district-level and municipal vaccination task forces to improve implementation and address challenges at the grassroots level, he said.
Municipal task forces, chaired by respective civic commissioners, have been constituted in view of the vast urban population in Maharashtra and the role of civic bodies in implementing different health programmes.
The district-level task forces will function under the chairmanship of collectors.
"Complete immunisation of children at the appropriate age is an extremely simple, cost-effective and highly effective measure to reduce child mortality and the prevalence of diseases among kids. Immunisation is a powerful tool for reducing illness in children," maintained the official.
To ensure full vaccination of all children, the state government implements various campaigns from time to time as per the central government guidelines, he pointed out.
"Active participation and cooperation of other relevant government departments are essential (in making these campaigns successful)," according to the official.
The state-level body will review the regular immunisation programme, associated campaigns and vaccine-preventable diseases in detail. It will also conduct focused assessments of high-risk districts and municipal corporations, including vacancies at district, municipal and sub-district levels, availability of cold chain equipment, resource gaps and training requirements, he noted.
The state task force will review allocation and utilisation of funds for immunisation and ensure timely action by officers concerned based on reports from district and municipal task forces and state-level monitoring mechanisms, the official said.
It will also ensure active coordination and participation of other government departments in immunisation drives, while district and municipal task forces will carry out similar functions at their respective levels, the official added.