New Delhi, Oct 15: The committee set up by the government to examine reports potentially linking the death of 66 children in The Gambia to four made-in-India cough syrups has observed that the clinical information shared by the World Health Organization so far is inadequate to determine the aetiology.
This was conveyed to the WHO by Drugs Controller General of India (DCGI) V G Somani on Saturday.
Rutendo Kuwana from the WHO on October 13 had written to the DCGI seeking to know the progress with the investigation of the manufacturer of the four cough syrups -- the Sonipat-based Maiden Pharmaceuticals.
In an email response, Dr Somani on Saturday said the Union Ministry of Health has constituted a committee of technical experts to examine and analyse the details of the adverse event reports and all related details shared by or to be shared by WHO and to recommend follow up action.
Somani said the four-member committee chaired by Dr Y K Gupta, Vice Chairperson, Standing National Committee on Medicines, in its first meeting has examined the reports and communications so far received from WHO and has made several observations.
Mentioning the observations, Dr Somani said, "The clinical features and the treatment received by the children as shared by WHO so far are inadequate to determine the aetiology."
The details of initial illness, sign and symptoms, duration of anura in the cases, results of laboratory investigations conducted including various markers and parameters, specific investigations for diethylene glycol and ethylene glycol on critical samples of the patients, treatment received before and after hospitalization at the tertiary hospital in Gambia, treatment received before and after acute kidney injury was suspected and reasons thereof, names and brands of the drug formulations used in the treatment before and after hospitalization, their manufacturers, their expiry other relevant information in each of the cases, are necessary, he said.
Dr Somani further said in case, verbal autopsy was conducted, a detailed report may be shared by WHO.
Dr Somani has sought details on the number of stool samples collected and analysed and the number of children with a similar clinical presentation to acute kidney injury from whom the biological samples were collected.
He also sought the details of the product samples collected and tested (with their compositions, manufacturers) which were reported to be negative for EG and DEG, the details of cases who received these products.
"You are therefore requested to share the report on causal relations along with above details, as observed by the said Technical Committee at the earliest for further examination and follow up action at our level," he wrote.
The DCGI further informed that the investigation of the manufacturer in question has progressed.
"The manufacturing site has been inspected, test reports of sampled products from the laboratory are awaited and all the manufacturing activities of the manufacturer in question have been stopped," he said.
The Union Health Ministry on Wednesday formed the four-member panel of experts to examine the details and adverse event reports received from the WHO on the deaths of 66 children in Gambia being linked potentially to the four cough syrups made by the Sonipat unit of Maiden Pharmaceuticals.
The committee will recommend further course of action to the Drugs Controller General of India (DCGI) after examining and analysing the adverse event reports, causal relationship and all related details already shared by or to be shared by the World Health Organization.
The causal relation of deaths is yet to be provided by WHO. The WHO has so far shared with CDSCO, the certificates of analysis of each of the sampled products, their photographs and summary of the adverse events.
WHO has stated that it is in process of further investigation, official sources said.
The CDSCO has already requested WHO to share at the earliest with it, the report on establishment of causal relation to death with the medical products in question and photographs of labels/products etc, they said.
While the Haryana State Drug Controller issued show cause notice to Maiden Pharmaceuticals on October 7 asking it to explain within a week "many contraventions" found during a recent inspection, or face suspension or cancellation of license, the Haryana government on October 11 issued an order to stop all the manufacturing activities of the firm with immediate effect.
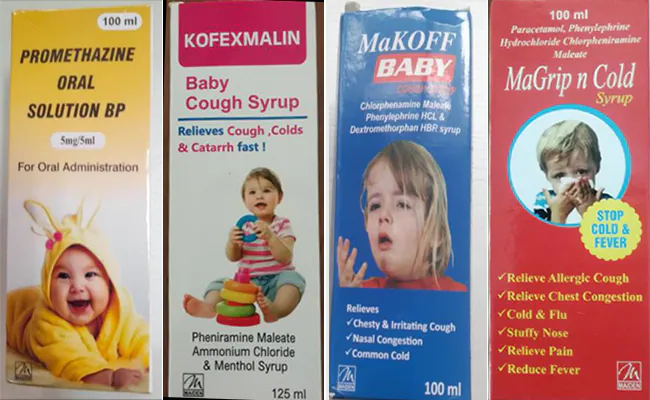
The WHO had on September 29 had informed the Central Drugs Standard Control Organisation (CDSCO) that they were providing technical assistance and advice to Gambia, where children have died and where a contributing factor was suspected to be the use of four medicines: Promethazine Oral Solution BP, KOFEXNALIN Baby Cough Syrup, MaKOFF Baby Cough Syrup and MaGrip n Cold Syrup.
They are manufactured and exported by Maiden Pharmaceuticals Ltd, Sonepat, Haryana, and the WHO had informed they may have been contaminated with Diethylene glycol or Ethylene glycol.
Let the Truth be known. If you read VB and like VB, please be a VB Supporter and Help us deliver the Truth to one and all.
New Delhi (PTI): India and Vietnam on Wednesday elevated their ties to an enhanced comprehensive strategic partnership and set a USD 25 billion annual trade target by 2030 as Prime Minister Narendra Modi and Vietnamese President To Lam held talks focusing on expanding bilateral ties in areas of trade, defence, and critical minerals in the face of increasing geopolitical upheavals.
Following the Modi-Lam meeting, the two sides signed as many as 13 agreements which will provide for deeper two ways engagements in a range of areas including digital payments, rare earth minerals, pharmaceuticals, education, banking and culture.
Modi and Lam also deliberated on the situation in the Indo-Pacific amid China's increasing assertiveness in the region, and agreed to continue to contribute to the rule-of-law, peace, stability, and prosperity.
It is understood that China's growing military muscle-flexing in the South China Sea also figured in the delegation-level talks between the two sides.
Elected as president earlier this month, Lam began his first state visit to India on Tuesday, leading a high-level delegation for a three-day trip.
ALSO READ: Explosives used in blast outside Punjab Frontier BSF HQ in Jalandhar: Punjab Police
"A decade ago, Vietnam became India's first comprehensive strategic partner in ASEAN (Association of Southeast Asian Nations) . Since then, our relations have made rapid and comprehensive progress," Modi said in his media statement.
"Building on this strong foundation, today we are elevating our relations to the level of Enhanced Comprehensive Strategic Partnership. We will now advance our partnership to even higher goals," he said.
"Our cooperation will reach new levels in every area ' culture, connectivity, and capacity building, as well as security, sustainability, and supply chain resilience," Modi added.
He noted that the bilateral trade between India and Vietnam has doubled in the last decade to reach USD 16 billion and that the target now is to increase it to USD 25 billion by 2030.
Modi also cited a plethora of agreements signed between the two countries to emphasis that the economic engagement is expected to witness a significant upswing going forward.
"The MoU between our drug authorities will now increase access to Indian medicines in Vietnam. Exports of Indian agricultural, fisheries, and animal products to Vietnam are also set to become easier. Very soon, Vietnam will taste India's grapes and pomegranates," he said.
The two sides also agreed to update the India-ASEAN trade agreement by the end of this year.
"This will provide a new impetus to trade and investment between India and all ASEAN countries. New initiatives in critical minerals, rare earths, and energy cooperation will ensure the economic security and supply chain resilience of both countries," Modi said.
"To boost financial connectivity, we have today decided to enhance cooperation between our central banks. India's UPI and Vietnam's fast payment systems will soon be linked. Furthermore, we are strengthening state-to-state and city-to-city cooperation between the two countries," he noted.
Modi said India is grateful to Vietnam for its strong condemnation of the Pahalgam terrorist attack and standing with India in its fight against terrorism.
"In this era of global turmoil and economic challenges, thanks to their talent, good governance, and economic reforms, India and Vietnam are emerging as the world's fastest-growing economies," he said.
"Now, through our enhanced strategic partnership, we will support each other's rapid growth. As the Buddha's teachings say, 'If you light a lamp for someone else, it also illuminates your own path'," Modi said.
He also described Vietnam a a key pillar of India's Act East Policy and Vision Ocean.
"In the Indo-Pacific region as well, we share a common outlook. Through our strengthening defence and security cooperation, we will continue to contribute to the rule-of-law, peace, stability, and prosperity," he said.
The prime minister said India will broaden its relations with ASEAN with Vietnam's cooperation.
In his remarks, Lam said both sides agreed to deepen political trust and elevate security cooperation.
Last year, the two sides inked a pact to set up a framework for submarine search, rescue and support mechanism. They also signed a letter of intent (LoI) to strengthen bilateral defence industry collaboration.