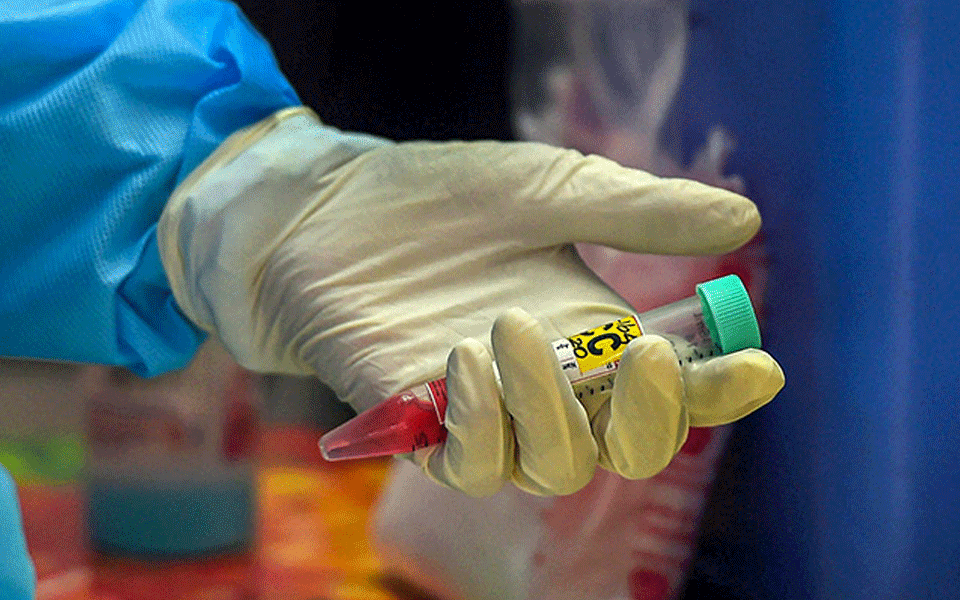
New Delhi: India's drug regulator has approved Itolizumab, a drug used to cure skin ailment psoriasis for "restricted emergency use" to treat COVID-19 patients with moderate to severe acute respiratory distress, officials told PTI on Friday.
Considering the unmet medical needs to treat COVID-19, Drugs Controller General of India, Dr V G Somani, approved monoclonal antibody injection Itolizumab, an already approved drug of Biocon, for restricted emergency use for the treatment of cytokine' release syndrome in moderate to severe acute respiratory distress syndrome patients due to COVID-19, they said.
"The approval was given after its clinical trials on COVID-19 patients in India was found satisfactory by the expert committee comprising pulmonologists, pharmacologists and medicine experts from AIIMS, among others, for treatment of cytokine release syndrome, an official told PTI.
"It is already an approved drug of Biocon for treating psoriasis for last many years," the official said.
Written informed consent of each patient is required before the use of this drug, he said.
Let the Truth be known. If you read VB and like VB, please be a VB Supporter and Help us deliver the Truth to one and all.
Gangtok (PTI): Prime Minister Narendra Modi on Tuesday launched development projects worth over Rs 4,000 crore in Sikkim while attending the closing ceremony of the state's 50 years of statehood celebrations.
The projects span a wide range of sectors such as infrastructure, connectivity, healthcare, education, power, urban development, environment, tourism and agriculture, the officials said.
These projects are aimed at accelerating holistic and inclusive development in Sikkim.
The PM's visit marks an important milestone in the golden jubilee year of Sikkim's statehood and reflects the government's "continued commitment to the rapid and sustainable development of the Himalayan state", the officials said.